 On the basis of such results, it is indicated that, as was found in earlier work, a thermodynamic theory of irreversible processes can be erected on the compensation function since it is an integral of a one‐form in the thermodynamic space whereas the Boltzmann entropy is not. Such a limit represents a contraction of information as the description of irreversible processes is made in the thermodynamic space contracted from the phase space of 10 23 particles. 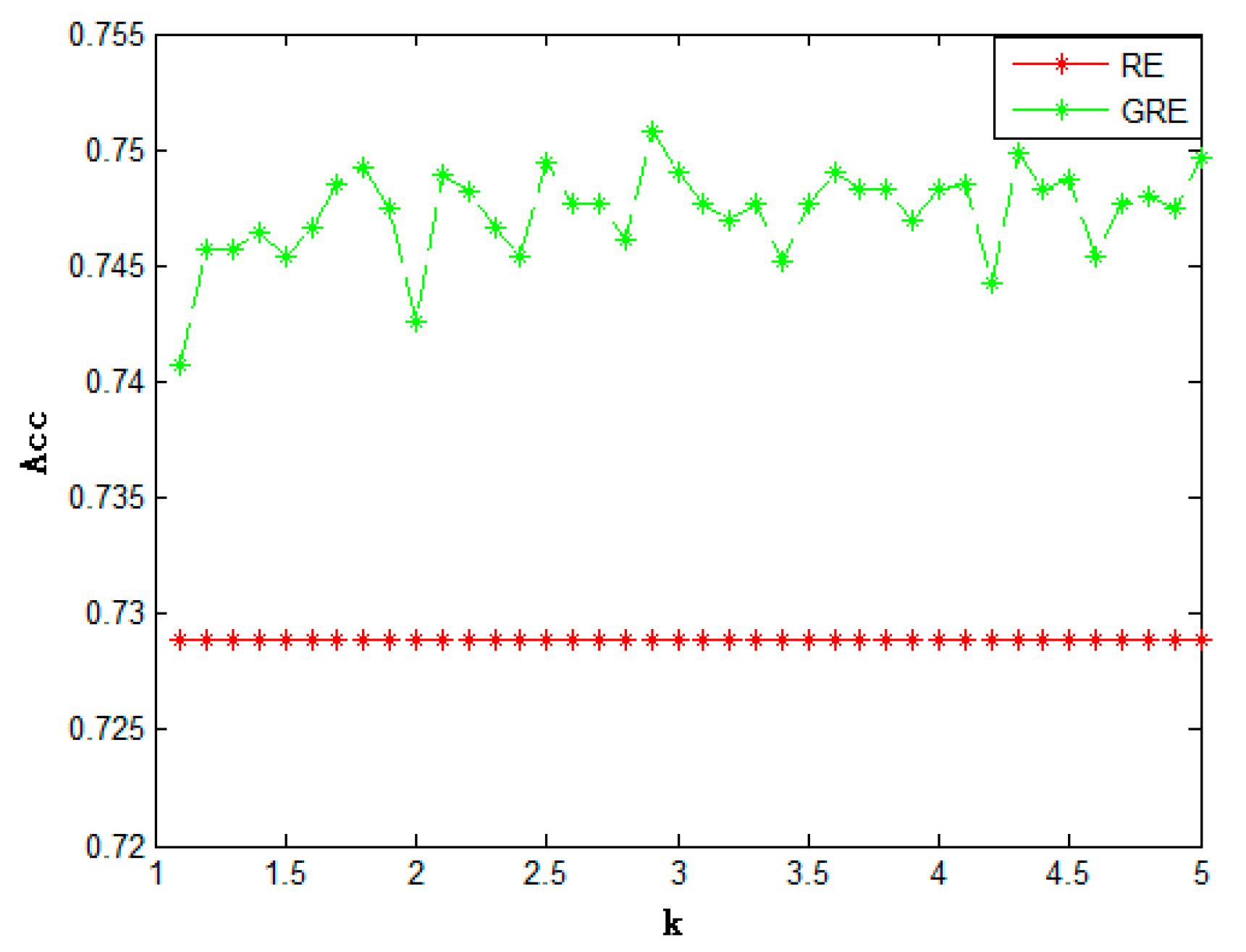
The time derivative of the relative entropy does not vanish in the limit, but tends to a limit associated with energy dissipation. freezing), or from a gas to a liquid (i.e. By using the balance equations for the compensation function and the relative entropy, we investigate the limiting behavior of the rate of relative entropy as the thermodynamic branch of the distribution function becomes convergent in the sense of means (i.e., weakly converges) to the phase‐space distribution function. A phase change from a liquid to a solid (i.e. By using the concept of relative entropy which is the difference between the two quantities, we examine their relations and significance for the mathematical structure of thermodynamics of irreversible processes. The two quantities are not generally the same. The unifying concept is of absolutely continuous measure. The Boltzmann entropy is an information entropy enumerated with the phase‐space distribution function in the phase space, whereas the compensation function introduced in the previous work is a representation of the former in thermodynamic space. I think you have introduced good ideas, but some care is needed to make sense of all this. Such a distribution function is different in nature from the phase‐space distribution function obtained by directly solving the Boltzmann kinetic equation subject to initial and boundary conditions in the phase space without the functional hypothesis. Formally, given two probability distributions p(x) and q(x) over a discrete random variable X, the relative entropy given by D(pjjq) is de ned as follows: D. In the 1940–1950’s the notion of entropy turned out to be central in information theory, a field pioneered by mathematicians such asĪssume we have a categorical distribution \(P\) with \(K\) classes/categories.The conventional solution methods for the Boltzmann kinetic equation such as the Chapman–Enskog method or the moment method provide a thermodynamic branch of the distribution function evolving through macroscopic variables under the functional hypothesis. 2 Relative Entropy The relative entropy, also known as the Kullback-Leibler divergence, between two probability distributions on a random variable is a measure of the distance between them. The probabilistic interpretation of statistical mechanics andĮntropy was further developed by J. 
Lemma 3.Jensen's Inequality:LetQdenote a function on a random variableX. The entropy is a property of the underlying distributionPU(u) u2 Uthat measures the amount ofrandomness or surprise in the random variable. 
The modern definition of (relative) entropy, or “disorder”, was first discovered in the 1870s by physicist L. 1 H(U),ES(U) ElogE p(U) log (p(U)) p(u) logp(u) (2) u WhereUrepresents all u values possible to the variable. Elaborating on a previous work by Marolf et al, we relate some exact results in quantum field theory and statistical mechanics to the Bekenstein universal bound on entropy. 
In this chapter we discuss various information criteria and their connection to maximum likelihood. Relative entropy and the Bekenstein bound. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
